Veterinary Medicines Directorate
475 articles
Showing 391-420

Supplementary protection certificates: IPO call for views
Thursday, 31 May 2018
Opportunity to comment on EU proposal to create an exception from infringement of supplementary protection certificates for medicines manufactured for...

Veterinary Medicines Directorate office closure for Bank Holiday
Friday, 25 May 2018
The VMD offices will be closed Monday 28 and Tuesday 29 May...

Suspension of Marketing Authorisations
Thursday, 24 May 2018
Details of the suspension of marketing authorisations relating to Bio-Tech Solutions Ltd and Mr H.I. & Mrs S.J. Moulds....

VMD announces the new Director of Authorisations: Abigail Seager
Wednesday, 25 April 2018
The new Director of Authorisations will take up her position on 1 May 2018....

The VMD Customer Satisfaction Survey 2018: Results
Tuesday, 03 April 2018
Results of the Customer Satisfaction Survey 2018....

Veterinary medicines: Changes to distributor and local representatives details
Thursday, 22 March 2018
We have changed the way we deal with variations to change distributor and local representative details....

VMD web and switchboard services will be unavailable from 16:00, 23 February to 8:00, 26 February
Thursday, 22 February 2018
Web services, such as the Special Imports System, and the switchboard service will be unavailable due to essential electrical maintenance. ...

VMD offices closing early on Friday 22 December
Thursday, 21 December 2017
Due to an electrical shut down the VMD offices will be closed from 13:45pm on Friday 22 December....

VMD: Civil Service People Survey 2017
Thursday, 07 December 2017
This highlights report provides details of how the Veterinary Medicines Directorate (VMD) performed in the Civil Service People Survey....

Animal medicines seizure: Mr M Priestly
Friday, 24 November 2017
Details of the seizure notice served on Mr M Priestly, Downpatrick, Northern Ireland....

The VMD Customer Satisfaction Survey 2017/18: Improving our service to you
Tuesday, 31 October 2017
The VMD launched its customer satisfaction web survey on 30 October 2017....

Pharmacovigilance system: Eudravigilance Downtime
Wednesday, 18 October 2017
The EMA Eudravigilance system will be unavailable from 8 November to 21 November 2017....

Veterinary Medicines Directorate: Authorisation application deadlines
Wednesday, 18 October 2017
Authorisation application deadlines for a MA, ManA, WDA, Batch Release and Export Certificate during the Christmas period 2017....

Startect Dual Active Oral Solution for Sheep: change of distribution category
Friday, 25 August 2017
Change of distribution category from POM-V to POM-VPS....

VPC and VMD Open meeting 2017
Friday, 04 August 2017
The Veterinary Products Committee and the Veterinary Medicines Directorate will hold their annual open meeting on Friday 29 September 2017....

VMD's Accredited Internet Retailer Scheme (AIRS) 5th Anniversary
Monday, 31 July 2017
Scheme that provides assurance to the public and professional keepers of animals when buying medicines online....

Veterinary Medicines Directorate Stakeholder Workshops
Wednesday, 26 July 2017
Stakeholder group workshops to discuss the implications of EU Exit to be held during the Autumn 2017....

VMD Pharmaceutical Industry Event Presentation 2017
Thursday, 13 July 2017
Veterinary Medicines Directorate Pharmaceutical Industry Event presentation to attendees on 23 June 2017. ...

Veterinary Medicines Containing Zinc Oxide: European Commission Decision
Wednesday, 05 July 2017
Details of the European Commission decision to withdraw the marketing authorisations of veterinary medicines containing zinc oxide for food producing ...

First veterinary DNA vaccine authorised for Clynav
Thursday, 29 June 2017
First DNA vaccine authorised in the EU to protect Atlantic salmon against Salmon Pancreas Disease....

VMD Pharmaceutical Industry Information Event 2017
Monday, 12 June 2017
Information event for the pharmaceutical industry on Friday 23 June 2017....
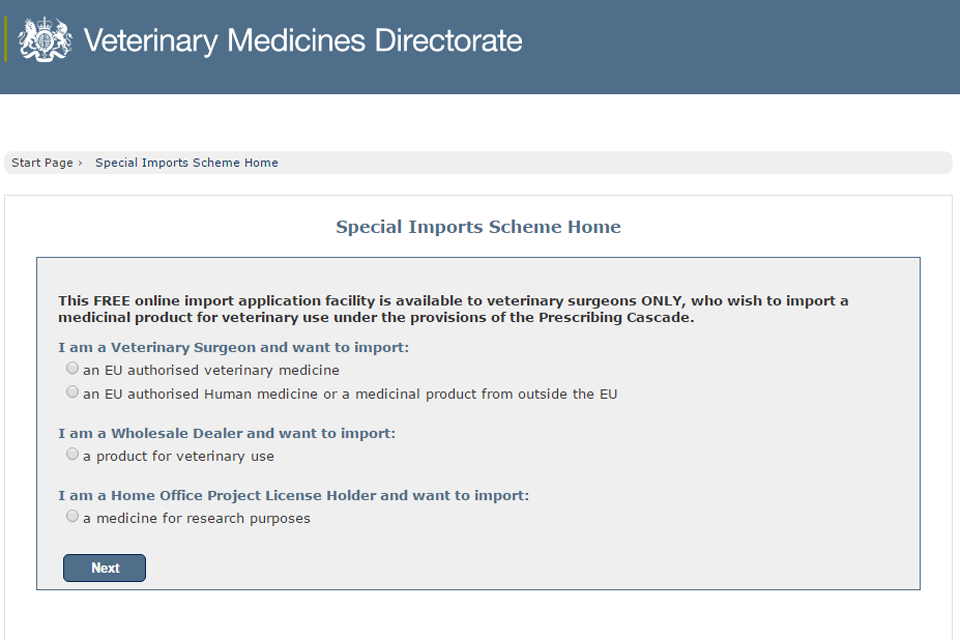
VMD web services will be unavailable from 27 to 31 May: Update 31 May 16:00pm
Wednesday, 31 May 2017
Web services, such as the Special Imports System, will be unavailable due to essential IT maintenance on the Bank Holiday weekend to 31 May 2017. ...

Veterinary Medicines Pharmacovigilance Annual Review 2015: Summary
Tuesday, 18 April 2017
A summary of results from surveillance work carried out by the VMD's pharmacovigilance team concerning reported adverse events....

Restrictions on Schedule 6 (Exemptions for small pet animals) active substances
Friday, 31 March 2017
Details of VMD review of the approved active substances on the exemption for small pet animals list which may be marketed for use as anaesthetics in f...

VMD on social media
Monday, 27 March 2017
We are now on Twitter and Facebook....
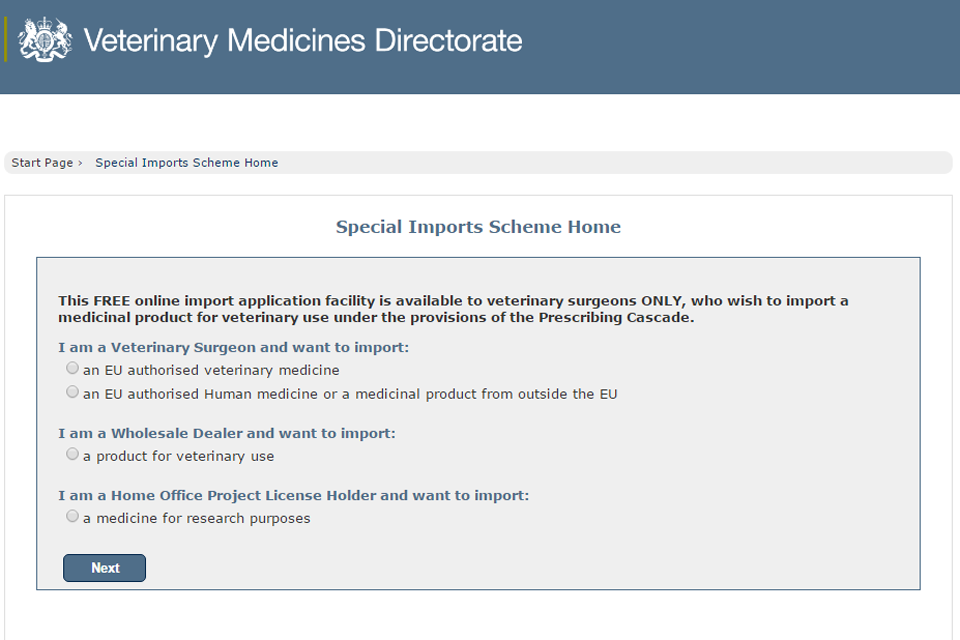
VMD web services will be unavailable on 25 March 2017
Wednesday, 22 March 2017
Web services, such as the Special Imports System, will be unavailable due to essential maintenance from 7am to 3pm on Saturday 25th March. ...

Veterinary medicines containing zinc oxide: European referral process
Monday, 20 March 2017
The EMA Veterinary Committee has reached a final conclusion that the benefit:risk balance for veterinary medicines containing zinc oxide is negative....

Zolvix 25 mg/ml Oral Solution for Sheep: change of distribution category
Tuesday, 14 March 2017
Change of distribution category from POM-V to POM-VPS...

European referral of veterinary medicines containing zinc oxide: Call for evidence
Monday, 06 February 2017
The EMA Veterinary Committee has concluded that the benefit:risk balance for veterinary medicines containing zinc oxide is negative and that this clas...

VMD on Twitter – What do you think?
Wednesday, 01 February 2017
Link to VMD survey...