Veterinary Medicines Directorate
475 articles
Showing 31-60

Launch of New Veterinary Medicines Packaging Surveillance Scheme
Monday, 05 January 2026
VMD introduces new surveillance scheme and updates mock-up assessment requirements to streamline regulation whilst maintaining standards. ...

Recruitment for a senior Policy Officer
Monday, 22 December 2025
Policy officer, working on policy and legislation on veterinary medicines....

Recruitment for a Service Desk Operator
Thursday, 18 December 2025
Service Desk Operator working as the first point of contact for IT support....

Recruitment for a Pharmaceutical Quality Assessor
Thursday, 18 December 2025
Pharmaceutical Quality Assessor working on marketing authorisations for veterinary medicines....

The UK and Canada agree to work together to improve regulatory efficiency for animal vaccines
Wednesday, 17 December 2025
UK and Canadian regulatory authorities have signed an agreement to enhance collaboration and improve access to animal vaccines....

Recruitment for a Policy Support Officer
Tuesday, 09 December 2025
Policy Support Officer, working on policy and legislation on veterinary medicines....

Recruitment for IT Operations Engineer (Infrastructure & Network Lead)
Tuesday, 02 December 2025
IT Operations Engineer (Infrastructure & Network Lead) who will lead the team responsible for the management of the VMD’s IT Infrastructure and Networ...

Webinars on changes to veterinary medicines supply into Northern Ireland from 1 January 2026
Tuesday, 02 December 2025
VMD has published webinars to help veterinary professionals, wholesalers and retailers prepare for changes to the supply of veterinary medicines into ...
DIVENCE IBR MARKER LIVE Lyophilisate and Solvent for Suspension for Injection (Cattle) SPC change
Monday, 01 December 2025
Change to the information provided in the Summary of Product Characteristics (SPC)....

Veterinary medicines regulator updates how it calculates adverse event incidence rates
Monday, 01 December 2025
VMD changes to calculation methods ensures figures reflect the way medicines are used....

Batch recall Anesketin 100 mg/ml Solution for Injection for Dogs, Cats and Horses
Friday, 28 November 2025
Batch recall alert for Anesketin 100 mg/ml Solution for Injection for Dogs, Cats and Horses by Dechra Pharmaceuticals PLC. ...

Recruitment for a Chief Financial Officer
Wednesday, 26 November 2025
Chief Financial Officer who will lead the team responsible for the management of the VMD’s finances....

Consultation on residues control programme charges
Monday, 24 November 2025
The VMD are consulting on proposed changes to charges for the National Residues Control Programme to recover full costs....

Authorisation application deadlines for Christmas period 2025
Friday, 21 November 2025
Validation for applications during the Christmas period 2025 for MA, ManA, WDA, Batch Release, Specific Batch Control and Export Certificates....

Recruitment for a Research Innovation Sustainability and Evidence Programme Coordinator
Wednesday, 19 November 2025
Research Innovation Sustainability and Evidence (RISE) Programme Coordinator, working on development and administration of projects across the RISE te...

Batch recall Kelactin and PGF Veyx Forte
Wednesday, 19 November 2025
Product defect recall alert for Kelactin 50 Microgram/ml Oral Solution for Dogs and Cats and PGF Veyx Forte 0.250mg/ml Solution for Injection for Catt...

Public and animal health benefit from record low sales of veterinary antibiotics
Tuesday, 18 November 2025
New report shows total quantity of antibiotics sold for livestock remains at the lowest level to date....

Batch recall alert Folltropin 700 IU Powder and Solvent for Solution for Injection
Friday, 14 November 2025
Product defect recall alert for Folltropin 700 IU Powder and Solvent for Solution for Injection by Vetoquinol SA and Vetoquinol UK Limited....

Veterinary medicine supply in Northern Ireland beyond 2025
Thursday, 13 November 2025
VMD provides details on schemes to support continued access to veterinary medicines from 1 January 2026....

Recruitment for a Business Relationship Manager
Tuesday, 04 November 2025
Business Relationship Manager, working on VMD’s alignment with Defra/government policies....

Batch recall alert for Vey Tosal 100 mg/ml + 0.05 mg/ml Solution for Injection
Monday, 03 November 2025
Product defect recall alert for Vey Tosal 100 mg/ml + 0.05 mg/ml Solution for Injection for Horses, Cattle and Dogs by Veyx-Pharma GmbH....

Recall alert Catophos 100 mg/ml + 0.05 mg/ml Solution for Injection
Thursday, 30 October 2025
Product defect recall alert for Catophos 100 mg/ml + 0.05 mg/ml Solution for Injection for Horses, Cattle and Dogs by Duggan Veterinary Supplies Limit...

Neptra Ear Drops Solution for Dogs Product defect recall alert
Thursday, 30 October 2025
Product recall alert for Neptra Ear Drops Solution for Dogs by Elanco Animal Health GmbH....

Recruitment for a Deputy Head of the Supply Team
Thursday, 23 October 2025
Deputy Head of the Supply Team, working on the Special Import Scheme....
Product defect recall alert for Busol and Procapen Injector
Wednesday, 22 October 2025
Recall alert for Busol 0.004 mg/ml Solution for Injection for Cattle, Horses, Rabbits and Procapen Injector 3g Intramammary Suspension for Cattle by ...
Product defect recall alert for four products by Dechra Pharmaceuticals PLC
Tuesday, 21 October 2025
Wholesaler level recall alert for four products by Dechra Pharmaceuticals PLC....

Small pet animal medicines exempt from authorisation: Annual submission of product list
Friday, 17 October 2025
Companies marketing medicines for small pet animals exempt from the requirements of a Marketing Authorisation must submit an annual return to the VMD ...

Tilmodil 300 mg/ml Solution for Injection for Cattle and Sheep Product defect recall
Thursday, 16 October 2025
Product recall alert for Tilmodil 300 mg/ml Solution for Injection for Cattle and Sheep by Emdoka & DUG(V) UK Limited. ...
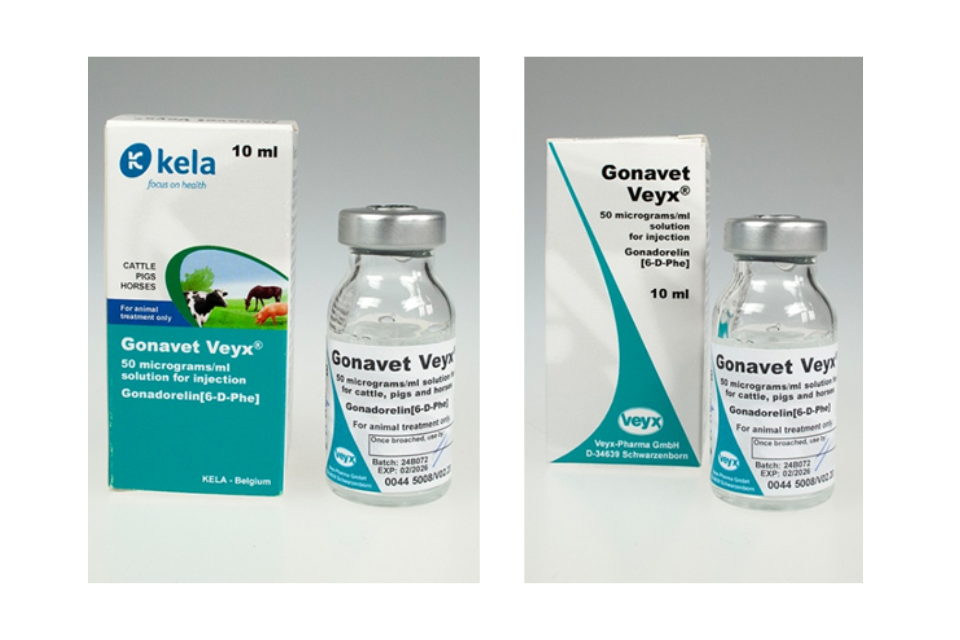
Gonavet Veyx 50 micrograms/ml Solution for Injection for Cattle, Pigs and Horses Product defect recall
Tuesday, 14 October 2025
Product recall for Gonavet Veyx 50 micrograms/ml Solution for Injection for Cattle, Pigs and Horses by Anupco Ltd T/A Kela Animal Health UK....
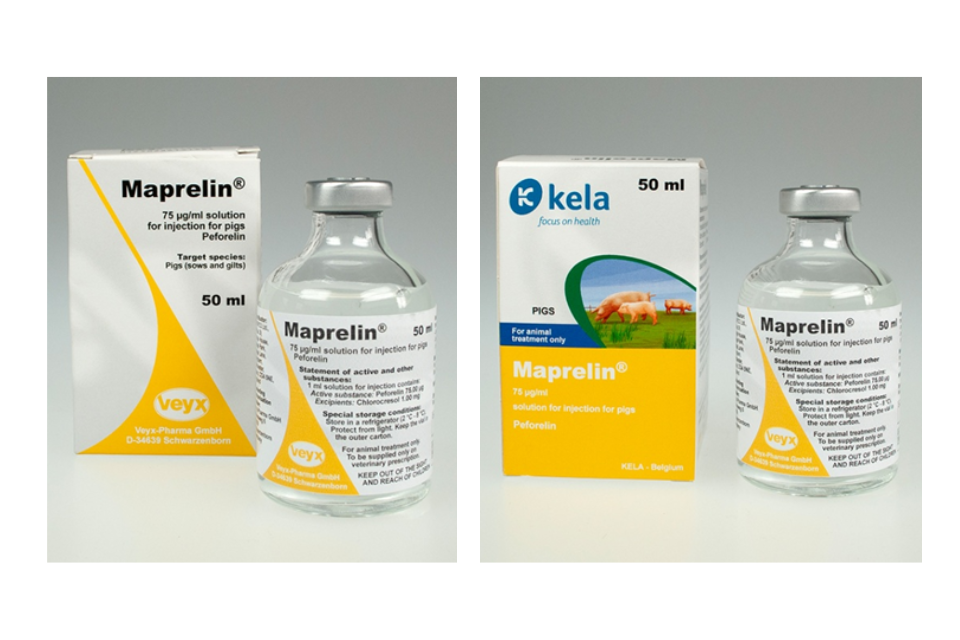
Maprelin 75 µg/ml Solution for Injection for Pigs Product defect recall
Tuesday, 14 October 2025
Product recall for Maprelin 75 µg/ml Solution for Injection for Pigs by Anupco Ltd T/A Kela Animal Health UK....